- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
02. October 2018
The formulation of amorphous solid dispersions (ASDs) is an effective way to improve the bioavailability of poorly water-soluble active pharmaceutical ingredients (APIs). The combination of an amorphous state of the drug and the presence of crystallization-inhibiting polymers retains a high amount of dissolved API over time. ASDs with ketoconazole and different polymers were manufactured by spray drying and their characteristics as well as performance were analyzed. Dissolution tests with a...
30. August 2018
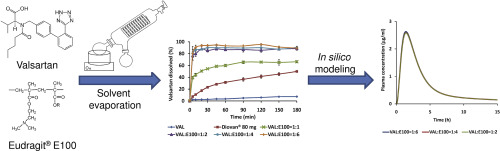
This study aimed to improve dissolution rate of valsartan in an acidic environment and consequently its oral bioavailability by solid dispersion formulation. Valsartan was selected as a model drug due to its low oral bioavailability (~23%) caused by poor solubility of this drug in the low pH region of gastrointestinal tract (GIT) and presence of absorption window in the upper part of GIT. Solid dispersions were prepared by solvent evaporation method with Eudragit® E100, Soluplus® or...
30. August 2018
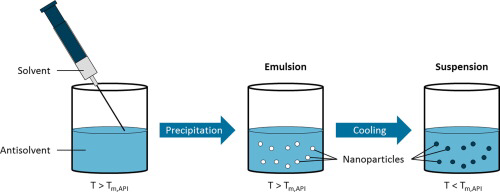
Antisolvent precipitation of poorly water-soluble drugs is a promising formulation technique to synthesize amorphous nanoparticles. The dissolution behavior of these nanoparticles is improved because of the high specific surface area and the amorphous state, leading to an enhanced bioavailability of the drug molecules. Nevertheless, stabilization of precipitated drug nanoparticles against agglomeration and recrystallization, which constitutes a key issue for further processing steps, has turned...
15. June 2018
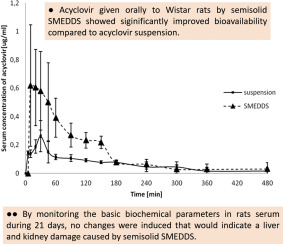
Semisolid self-microemulsifying drug delivery system (SMEDDS) with optimized drugloading capacity, stability, dispersibility in aqueous media and invitro drug release profile, was evaluated in vivo regarding effects on pharmacokinetics of acyclovir, an antiviral with low bioavailability (BA) and short half-life (t1/2). Additional goal of this study was evaluation of safety of this semisolid SMEDDS consisted of medium chain length triglycerides (oil) (10% w/w), macrogolglycerol hydroxystearate...
06. June 2018
Hot-melt extrusion is well established in the plastics industry and is applied in pharmaceutical application for more than 30 years. In contrast to plastics, pharmaceutical formulation development is more complex as each formulation is unique in terms of compositions of active pharmaceutical ingredients, carrier and additives. Therefore, new innovative solutions are demanded to tackle the pharma specific challenges in formulation development like how to prevent losses with expensive material...
05. June 2018
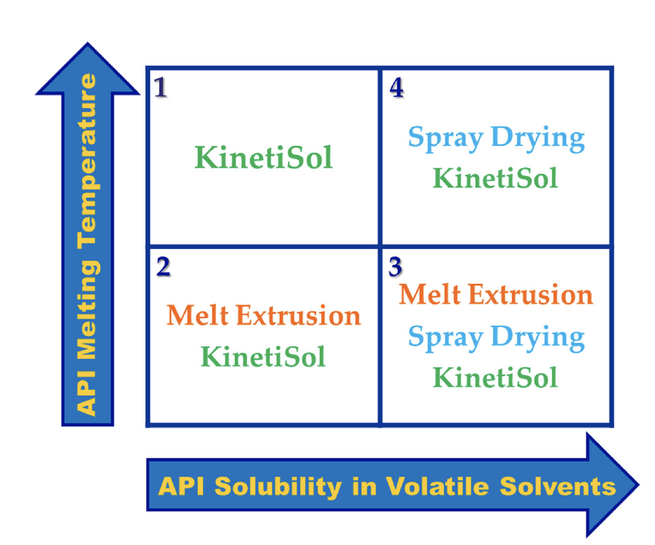
Due to the high number of poorly soluble drugs in the development pipeline, novel processes for delivery of these challenging molecules are increasingly in demand. One such emerging method is KinetiSol, which utilizes high shear to produce amorphous solid dispersions. The process has been shown to be amenable to difficult to process active pharmaceutical ingredients with high melting points, poor organic solubility, or sensitivity to heat degradation. Additionally, the process enables classes...
04. June 2018
Complex hydrogels formed with chitosan (CS) and ring-opened polyvinyl pyrrolidone (roPVP) as a swellable mucoadhesive gastroretentive drug dosage form (smGRDDF) were prepared and characterized. CS/roPVP hydrogels were produced by blending CS with roPVP obtained by basic treatment of PVP. Effects of the heating time and NaOH concentration employed for preparing roPVP, and CS molecular weights (Mws), and roPVP/CS ratios on the swelling ability of the resultant hydrogels were characterized....
30. May 2018
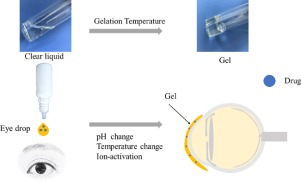
Blindness and vision impairment are the most devastating global health problems resulting in a substantial economic and social burden. Delivery of drug to particular parts of the anterior or posterior segment has been a major challenge due to various protective barriers and elimination mechanisms associated with the unique anatomical and physiological nature of the ocular system. Drug administration to the eye by conventional delivery systems results in poor ocular bioavailability (< 5%)....
26. May 2018
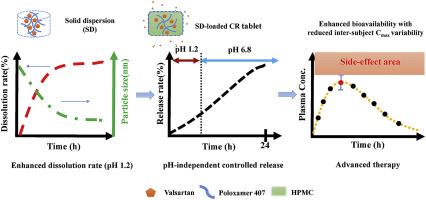
The aims of this work were to design pH-independent controlled release (CR) tablet containing nanonizing solid dispersion (SD) adsorbed on hydrophilic silica (Aeroperl® 300/30). Valsartan (VAL) was chosen to simultaneously modulate solubility and release rate due to its poor water solubility in low pH condition and short elimination half-life. Based on extensive equilibrium solubility and compatibility studies, poloxamer 407 was selected as a SD carrier. The melted mixtures of drug and...
24. May 2018
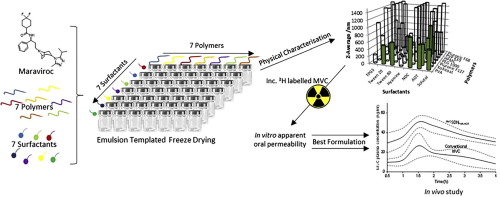
Oral drug administration remains the preferred approach for treatment of HIV in most patients. Maraviroc (MVC) is the first in class co-receptor antagonist, which blocks HIV entry into host cells. MVC has an oral bioavailability of approximately 33%, which is limited by poor permeability as well as affinity for CYP3A and several drug transporters. While once-daily doses are now the favoured option for HIV therapy, dose-limiting postural hypotension has been of theoretical concern when...