- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
24. February 2018
To investigate the nature of drug-excipient interactions between indomethacin (IMC) and methacrylate copolymer Eudragit® E (EE) in the amorphous state, and evaluate the effects on formulation and stability of these amorphous systems. Methods Amorphous solid dispersions containing IMC and EE were spray dried with drug loadings from 20% to 90%. PXRD was used to confirm the amorphous nature of the dispersions, and DSC was used to measure glass transition temperatures (Tg). 13C and 15N solid-state...
06. November 2017
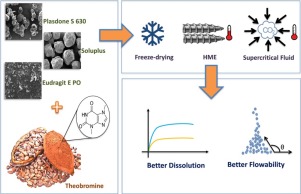
The aim of this study was to improve the pharmaceutical properties of theobromine (TB), particularly its flowability and dissolution rate, by preparing solid dispersions using different technologies (hot melt extrusion—HME, freeze-drying—FD, and supercritical fluid—SF) as well as testing different hydrophilic polymeric matrixes (Eudragit™ E, Plasdone™ S and Soluplus™)
06. July 2016
Abstract The phase transition of pharmaceutical excipients that can be induced by humidifying or heating is well-known to increase the hardness of orally disintegrating tablets (ODTs). However, these conditions are not applicable to drug substances that are chemically unstable against such stressors. Here, we describe a system which enhances the hardness of tablets containing water-insoluble polymers by using high-pressure carbon dioxide (CO2). On screening of 26 polymeric excipients,...
08. June 2015
The STEP stands for Safety and Toxicity of Excipients for Paediatrics. The Safety and Toxicity of Excipients for Paediatrics (STEP) Database is a user-designed resource developed by European Paediatric Formulation Initiative (EuPFI) and United States Paediatric Formulation Initiative (USPFI) in collaboration for storage and rapid/effortless access to the safety and toxicological data of excipients that is scattered over various sources and presents it in one freely accessible source. It is a...