- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
17. February 2018
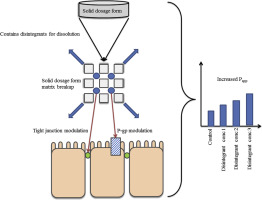
Abstract Pharmaceutical excipients were designed originally to be pharmacologically inert. However, certain excipients were found to have altering effects on drug pharmacodynamics and/or pharmacokinetics. Pharmacokinetic interactions may be caused by modulation of efflux transporter proteins, intercellular tight junctions and/or metabolic enzyme amongst others. In this study, five disintegrants from different chemical classes were evaluated for P-glycoprotein (P-gp) related inhibition and tight...
19. July 2017
The pharmaceutical excipient, polyethylene glycol 400 (PEG 400), unexpectedly alters the bioavailability of the BCS class III drug ranitidine in a sex-dependent manner
02. March 2017
Zidovudine (AZT) mucoadhesive solid dispersions (SD) were prepared using a sodium starch glycolate (SSG) and hypromellose phthalate (HPMCP) mixtures as carrier to enhance the intestinal permeability and bioavailability of zidovudine. SDs were prepared using the co-precipitation method followed by solvent evaporation and characterized according to their physicochemical properties such as particle size, crystallinity, thermal behavior, and liquid uptake ability. In vitro drug dissolution,...
11. October 2016
Abstract In this present study, the secretory transport of P-gp substrates, rhodamine 123 and digoxin, was evaluated using a Caco-2/HT29-MTX co-culture characterized by an efflux mechanism and a paracellular permeability closer to the human intestinal barrier compared to the Caco-2 monolayer gold standard. The influence of simulated intestinal fluids termed FeSSIF and FaSSIF on the intestinal absorption was also assessed in comparison with a conventional saline buffer. Labrasol® ALF and...
17. April 2016
Males and females respond differently to drugs: indeed, sex plays a crucial role in determining drug pharmacokinetics and pharmacodynamics. Excipients have also been shown to enhance the biovailability of drugs differently in men and women. The aim of this work was to investigate whether rodents are a good model in which to study sex-specific effects of polyethylene glycol 400 (PEG 400) on the bioavailability of ranitidine. Ranitidine (50 mg/Kg) was dissolved in water with different amounts of...
21. October 2015
Nowadays exploration of novel lipid-based formulations is akin to a magnet for researchers worldwide for improving the in vivo performance of highly lipophilic drugs. Over the last few years, new compositions of lipids have been developed, and the probable bioavailability enhancement has been investigated. We reviewed the most recent data dealing with backlogs of conventional lipid-based formulations such as physical instability, limited drug loading capacities, drug expulsion during storage...