- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
25. April 2018
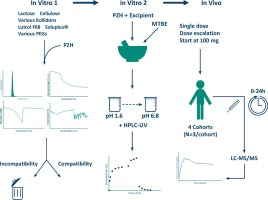
The anti-cancer drug pazopanib hydrochloride (PZH) has a very low aqueous solubility and a variable oral bioavailability. A new pharmaceutical formulation with an improved solubility may enhance the bioavailability and reduce the variability. A broad selection of polymer excipients was tested for their compatibility and solubilizing properties by conventional microscopic, thermal and spectrometric techniques. A wet milling and mixing technique was used to produce homogenous powder mixtures. The...
09. April 2018
Some impressions of the 2018 PBP Worldmeeting on Pharmaceutics, Biopharmaceutics and Pharmaceutical Technology in Granada with over 1.300 participants. 4 parallel session on hot topics, poster session and accompanying Exhibition by ResearchPharm allowed intense exchange amongst the participipants. More Information on the event
18. July 2016
OBJECTIVES: The aim of this study was to determine the influence of non-ionisable excipients hydroxypropyl-β-cyclodextrin (HPβCD) and poloxamers 407 and 188 on the supersaturation and precipitation kinetics of ibuprofen, gliclazide, propranolol and atenolol induced through solution pH shifts using the CheqSol method. METHODS: The drug's kinetic and intrinsic aqueous solubilities were measured in the presence of increasing excipient concentrations using the CheqSol method. Experimental data...
02. March 2015
Drug Targeting Liposomes and Nanoparticles Oral Drug Delivery Pulmonary Drug Delivery Skin / Vaginal / Rectal / Nasal / Ear / Ocular Drug Delivery Smart Drug Delivery Systems Transdermal Drug Delivery Vaccine Delivery http://www.europeanmeeting.org/home/