- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
26. August 2018
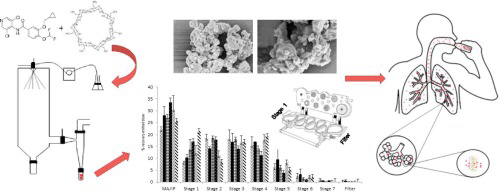
The aim of this study was to develop roflumilast dry powder inhaler (DPI) formulations by spray drying using hydroxypropyl-β-cyclodextrin (HPβCD) and to determine their suitability for pulmonary delivery. Different feed solution concentrations, solvent systems and spray drying parameters were used to obtain the formulations which were characterized using X-ray powder diffraction, thermal analysis, scanning electron microscopy, particle size distribution, bulk and tapped density, specific...
28. September 2017
Respiratory diseases such as asthma, chronic obstructive pulmonarydisease (COPD), and idiopathic pulmonary fibrosis are among the most serious and widespread healthcare challenges facing the developed world, accounting for more than 400,000 deaths in the EuropeanUnion, equivalent to 8% of the total mortality figure (1). Such figures highlight the growing need for innovative and effective treatments. Pulmonary drug delivery is receiving increased attention asa non-invasive means for local...
27. February 2017
Abstract Micronized cocrystal powders and amorphous spray-dried formulations were prepared and evaluated in vivo and in vitro as pulmonary absorption enhancement formulations of poorly soluble itraconazole (ITZ). ITZ cocrystals with succinic acid (SA) or l-tartaric acid (TA) with a particle size diameter of <2 μm were successfully micronized using the jet-milling system. The cocrystal crystalline morphologies observed using scanning electron microscopy (SEM) suggested particle shapes that...
09. February 2017
Abstract The effects of propellant type, cosolvent content, and air humidity on the morphology and solid phase of the particles produced from solution pressurized metered dose inhalers containing the corticosteroid beclomethasone dipropionate were investigated. The active ingredient was dissolved in the HFA propellants 134a and 227ea with varying levels of the cosolvent ethanol and filled into pressurized metered dose inhalers. Inhalers were actuated into an evaporation chamber under controlled...
07. January 2017
ABSTRACT: Bosentan is an endothelial receptor antagonist (ERA) indicated for the treatment of Pulmonary arterial hypertension (PAH). The aim of the present study involves the development of controlled release tablets of bosentan1. The tablets were prepared to release the drug for a prolong period of time within the GIT, to enhance the bioavailability, to minimize the dosing frequency, and to improve the patient compliance2. The tablets were formulated by using various polymers like hydroxyl...
13. December 2016
Abstract Previously, granulated lactose carriers were shown to improve uniformity and aerosolization of a low-dose model drug. In the present study, the blending uniformity and aerosol dispersion performance were assessed for 2 model drugs salbutamol sulfate (SS) and rifampicin (RIF), blended at high loadings (10% or 30% drug) with granulated lactose carriers. The model drug powders differed in particle size distribution, morphology, density, and surface energies. Content uniformity of RIF...
20. November 2016
Abstract Associating protein with nanoparticles is an interesting strategy to improve their bioavailability and biological activity. Solid lipid nanoparticles (SLN) have been sought as carriers for therapeutic proteins transport to the lung epithelium. Nevertheless, because of their low inertia, nanoparticles intended for pulmonary application usually escape from lung deposition. To overcome this problem, the production of spray-dried powders containing nanoparticles has been recently reported....
18. October 2016
Abstract The present study focuses on the development of dry powders for inhalation as adjuvant chemotherapy in lung cancer treatment. Cisplatin was chosen as a potential candidate for a local treatment as it remains the main platinum component used in conventional chemotherapies, despite its high and cumulative systemic toxicities. Bulk cisplatin was reduced to submicron sizes using high-pressure homogenization, mixed with a solubilized lipid and/or PEGylated component and then spray-dried to...
10. July 2016
Abstract The current work outlines the application of an up-to-date and regulatory-based pharmaceutical quality management method, applied as a new development concept in the process of formulating dry powder inhalation systems (DPIs). According to the Quality by Design (QbD) methodology and Risk Assessment (RA) thinking, a mannitol based co-spray dried formula was produced as a model dosage form with meloxicam as the model active agent. The concept and the elements of the QbD approach...
11. February 2016
Purpose: Local therapy is a valuable and strategic approach in the treatment of lung associated diseases and dry powder inhalation (DPI) formulations play the key role in this plan. Transfersome has been introduced as a novel biocompatible vesicular system with potential for administration in pulmonary drug delivery. The present study was designed to prepare Itraconazole-loaded nanotrantransfersomal DPI formulation. Methods: Itraconazole-loaded nanotransfersomes with three different types of...