- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
30. April 2018
The overall objective of this work is to understand how excipient characteristics influence the drug product quality attributes and process performance of a continuous twin screw wet granulation process. The knowledge gained in this study is intended to be used for Quality by Design (QbD)-based formulation design and formulation optimization. Three principal components which represent the overarching properties of 8 selected pharmaceutical fillers were used as factors, whereas factors 4 and 5...
31. March 2018
While some drug innovations are the result of surprises during research phases, a surprise that occurs during development--such as formulation or manufacturing problems--could result in approval delays or failure for a drug product.
To reduce the risk of late-phase surprises, some experts recommend that additional screening efforts in early development can smooth the pathway in later development stages. Developers of promising compounds emerging from drug discovery must balance the need to bett
18. August 2017
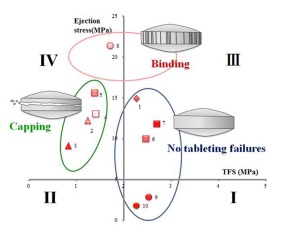
When tablet formulations are designed, it is necessary to understand “Tableting properties” and to determine the optimum type, grade, and amount of ingredients. “Tableting properties” consist of “Compressibility”, “Compactability”, and “Manufacturability”.