- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
18. March 2016
The aim of this study was to develop, evaluate and compare extended release mini-matrices based on metoprolol tartrate (MPT) and either glyceryl behenate (GB) or glyceryl palmitostearate (GPS). Mini-matrices were produced by three different techniques: hot melt extrusion, compression of melt granulates and prilling. Hot-melt extrusion and compression of granules obtained from melted material proved to be reliable, robust and reproducible techniques with aim of obtaining extended release...
05. February 2016
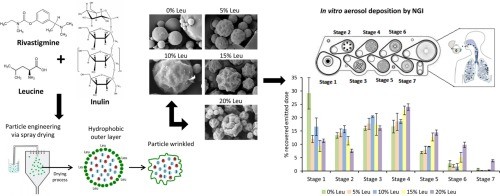
The purpose of this study was to prepare engineered particles of rivastigmine hydrogen tartrate (RHT) and to characterize the physicochemical and aerodynamic properties, in comparison to a lactose carrier formulation (LCF). Microparticles were prepared from ethanol/water solutions containing RHT with and without the incorporation of L-leucine (Leu), using a spray dryer. Dry powder inhaler formulations prepared were characterized by scanning electron microscopy, powder X-ray diffraction, laser...
13. January 2016
Ethylcellulose is one of the most commonly used polymers to develop reservoir type extended release multiparticulate dosage forms. For multiparticulate extended release dosage forms, the drug release is typically governed by the properties of the barrier membrane coating. The ICH Pharmaceutical Development Guideline (ICH Q8) requires an understanding of the influence of critical material attributes and critical process parameters on the drug release of a pharmaceutical product. More
06. October 2015
The aim of the present study was to evaluate the solid state characteristics, drug release and stability of fatty acid-based formulations after processing via prilling and solid lipid extrusion. Myristic acid (MA), stearic acid (SA) and behenic acid (BA) were used as matrix formers combined with metoprolol tartrate (MPT) as model drug. More
20. May 2015
A novel self-developed technique ultra-fine particle process system (UPPS) which is quite different from existing technology was employed to prepare microspheres for oral administration. The obtained MT microspheres by UPPS were spheroidal solid particles in size of about 80-120 μm with good flowability, which were fit for tableting or capsulation. The in vitro release behavior of the MT microspheres was similar with commercial sustained release tablet of metroprolol succinate, Betaloc®, and...