- Home
- Blog
- News
- Basics
- Sources
- Agencies, Regulatory & Organisations
- CERSI Excipients Browser
- Excipient Report
- Excipient DMF List
- EXCiPACT Certified Companies
- Excipient Documentation
- Excipient EINECS Numbers
- Excipient E-Numbers
- FDA Inactive Ingredient List
- FDA GRAS Substances (SCOGS) Database
- IPEC Americas
- USP - U.S. Pharmacopeia
- Definitions
- Whitepapers / Publications
- Supplier
- Services
- Media
- Events
- 1st pharmaexcipients Poster Award
- Event Calendar
- Events featured by pharma-excipients
- 4th Annual Formulation & Drug Delivery Congress
- DDF Summit
- ExcipientFest Americas
- ExcipientFest Asia
- Global CompliancePanel
- International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems
- Formulation & Drug Delivery USA Congress
- Laboratory Medicine 2018
- Making Pharmaceuticals Europe
- Making Pharmaceuticals Exhibition
- Pharma Integrates
- PharmaExcipients China @CPhI China
- TTC Technology Training Center
- Jobs
- Online Sourcing
- Contact
21. June 2018
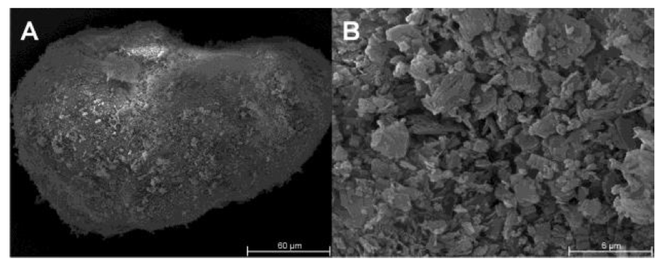
Tricalcium citrate (TCC) was characterized as a tableting excipient for direct compression (DC) and dry granulation (DG). SIGNIFICANCE: Brittle materials usually lead to tablets of inferior mechanical strength compared to plastic deforming materials. A brittle material exhibiting a high tabletability with the ability to retain that behaviour during recompression would represent a valuable alternative to the commonly used microcrystalline cellulose (MCC). METHODS: Tablets of TCC and other common...
12. February 2018
There is considerable interest in formulations with high active pharmaceutical ingredient (API) load, for reasons including lower patient tablet burden and therefore, potentially improved patient adherence. This remains a challenge not least because most APIs are poor flowing.
14. December 2017
Dow Pharma Solutions demonstrated its commitment to advancing the pharmaceutical sciences by exhibiting research findings in a range of posters at the 2017 AAPS show in San Diego, CA. Please find below links to copies of our presented posters.
17. October 2017
ARMOR PHARMA™ lactose monohydrate 350M is a fine milled powder of α-lactose monohydrate.
Higly compressible, this grade is mostly intended to be used in tablets formulation by Wet or Dry granulation.
11. September 2017
ARMOR PHARMA - technological lactose excipients - currently markets 3 types of pharmaceutical lactose.
ARMOR PHARMA™ lactose monohydrate : sieved & milled lactose
EXCIPRESS™ lactose for Direct Compression
EXCIPURE™ lactose for Dry powder Inhalation
26. August 2017
We aimed to investigate the feasibility of our previously proposed method for improving the properties of dry granules by pre-coating of active pharmaceutical ingredients (API) with a submicron-sized binder before conventional roller compaction in lab-scale production.
12. June 2017
A new ‘assisted' dry granulation method has been devised for the twin-screw granulator.